Carbon and Silicon may be together, but then there's a jump on the next ring to Germanium and Tin, and another jump to Lead and Flerovium. This also works for the transition metals, lanthanides and actinides, because their positions reflect the nature of the d and f orbitals.Īlso, noble gases aside, working out which element is in which group is nigh impossible using the Mayan Periodic Table. For example, the first row contains the atoms which only have 1s electrons, the second row fills up the 2s and 2p orbitals, the third row fills up the 3s and 3p orbitals, and so on. What the Periodic Table does well is it separates the elements according to how their orbitals are filled up. RE: The 'Mayan' Periodic Table - Pyrite - 03-16-2019 (I guess they're trying to keep it by using different colours to correspond to columns in the original table however, it is pretty messy ) The relations for the Group I metals and the halogens still kind of work, since those are all still sort of in a column however, beyond that, any semblance of neat 'columns' does fall apart on this thing. 
Well, any relations that apply to rows would still work: it's just that they would apply to rings instead. RE: The 'Mayan' Periodic Table - Kyng - 03-16-2019 It makes little sense to put the most none reactive (noble gases, group 18) next to the most reactive (group 1) These relations are now lost with the circle.Īs far as the noble gasses being "Group 0" that was based on incorrect thinking that there was no way a noble gas could combine electrons with other elements, and why if they are refereed to a "group" now its "Group 18". noble gasses, or how the first row sans H, reacts with water more and more violently as you go down) Rows also have significant too which I have forgotten. I only took Chemistry in high school but I do see one flaw in it, From what I recall, elements in the same column share certain properties (e.g. RE: The 'Mayan' Periodic Table - Lurkerish Allsorts - 03-16-2019 (However, this rule of "closer to the top = more reactive" falls apart when you consider the noble gases themselves: they're at the top, so by this rule they should be the most reactive, but in fact, they're completely inert )Īnyway, what are your thoughts on this table? 
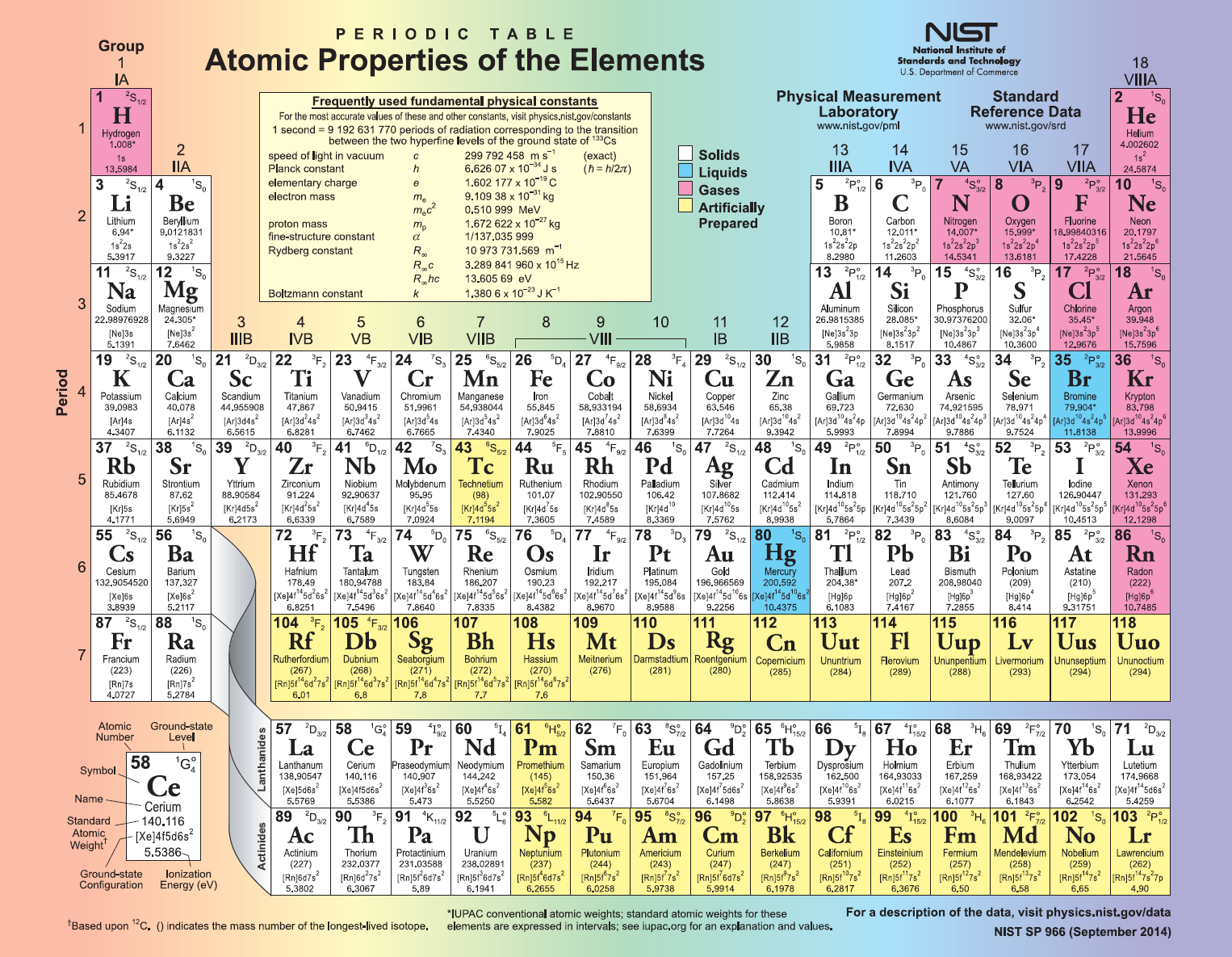
Another advantage is that it does a good job of showing the reactivity of the elements: the most reactive ones are those closest to the noble gases at the top of the table. For example, it does a better job of showing the lanthanides and actinides: on the traditional periodic table, these have to be shown in their own separate block, but in the Mayan table, they can be shown together with the others (since there's more space available the further out you go). This arrangement has certain advantages over the traditional periodic table. Then, the Group 1 elements are to the right of these the Group 2 elements are to the right of these, and so on (so, you can think of it as being a bit like a clock). The gold line at the top of the Mayan table contains the noble gases (which are often considered to be 'Group 0' of the traditional periodic table). In much the same way that the standard Periodic Table is composed of seven rows (or 'periods'), the Mayan table is composed of seven rings: each ring of the Mayan table corresponds to a row on the standard table. One alternative is the following circular version, known as the 'Mayan' Periodic Table, due to its visual similarity to Mayan calendars: (Click to enlarge) (Source: Mayan Periodic View licence terms) However, this certainly isn't the only way in which the 118 known elements can be depicted. We're all familiar with the traditional Periodic Table of the elements, which organises the elements into rows and columns: (Source: Offnfopt)įor over a century now, this has been the standard method for depicting the chemical elements, which form the basic building blocks of everything we encounter in our daily lives. The 'Mayan' Periodic Table - Kyng - 03-16-2019 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
